Claris’ Lead Candidate
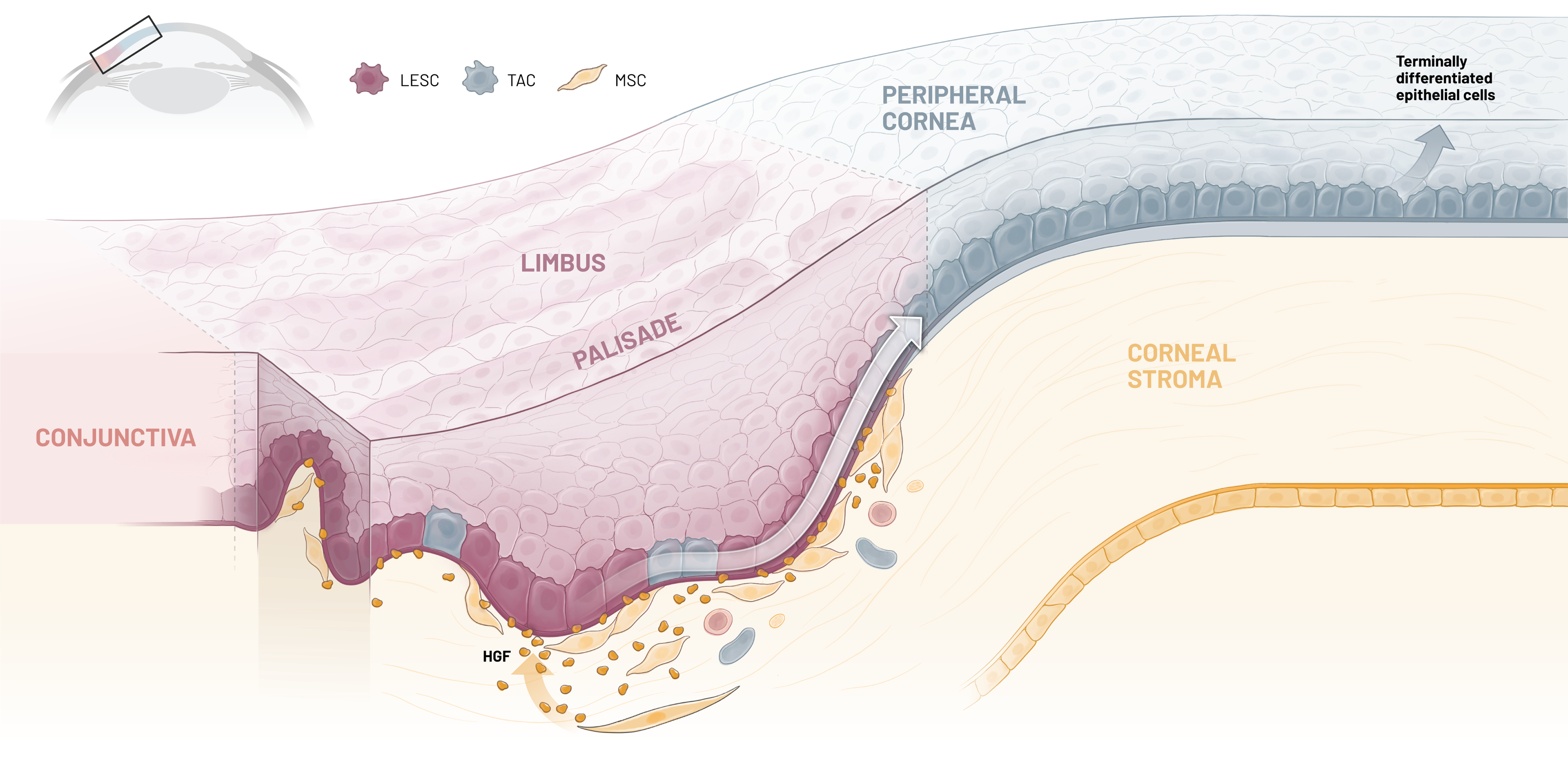
Our lead candidate, CSB-001, is a recombinant human HGF therapy developed to address the underlying pathophysiology of limbal stem cell deficiency (LSCD). In promoting corneal epithelial regeneration while reducing inflammation and fibrosis, CSB-001 has been shown to support cell survival by targeting the fundamental biological drivers of disease.
Designed as a topical ophthalmic therapy, CSB-001 has the potential to offer a reliable, accessible, and scalable treatment option that reduces reliance on complex surgical interventions and long-term immune suppression.

A Multi-Modal Therapeutic Approach
CSB-001’s broad biological activity engages multiple molecular pathways relevant to LSCD. By supporting limbal epithelial stem cell function, CSB-001 is designed to increase corneal epithelial regeneration while delivering anti-inflammatory, anti-fibrotic, and pro-cell-survival activity.
CSB-001 is designed to support earlier intervention in the disease course, expand access beyond specialized centers, and enable long-term disease management. In doing so, CSB-001 has the potential to reduce reliance on limbal grafting and other complex surgical procedures that are resource-intensive, variable in outcome, and inaccessible to many patients.
CSB-001
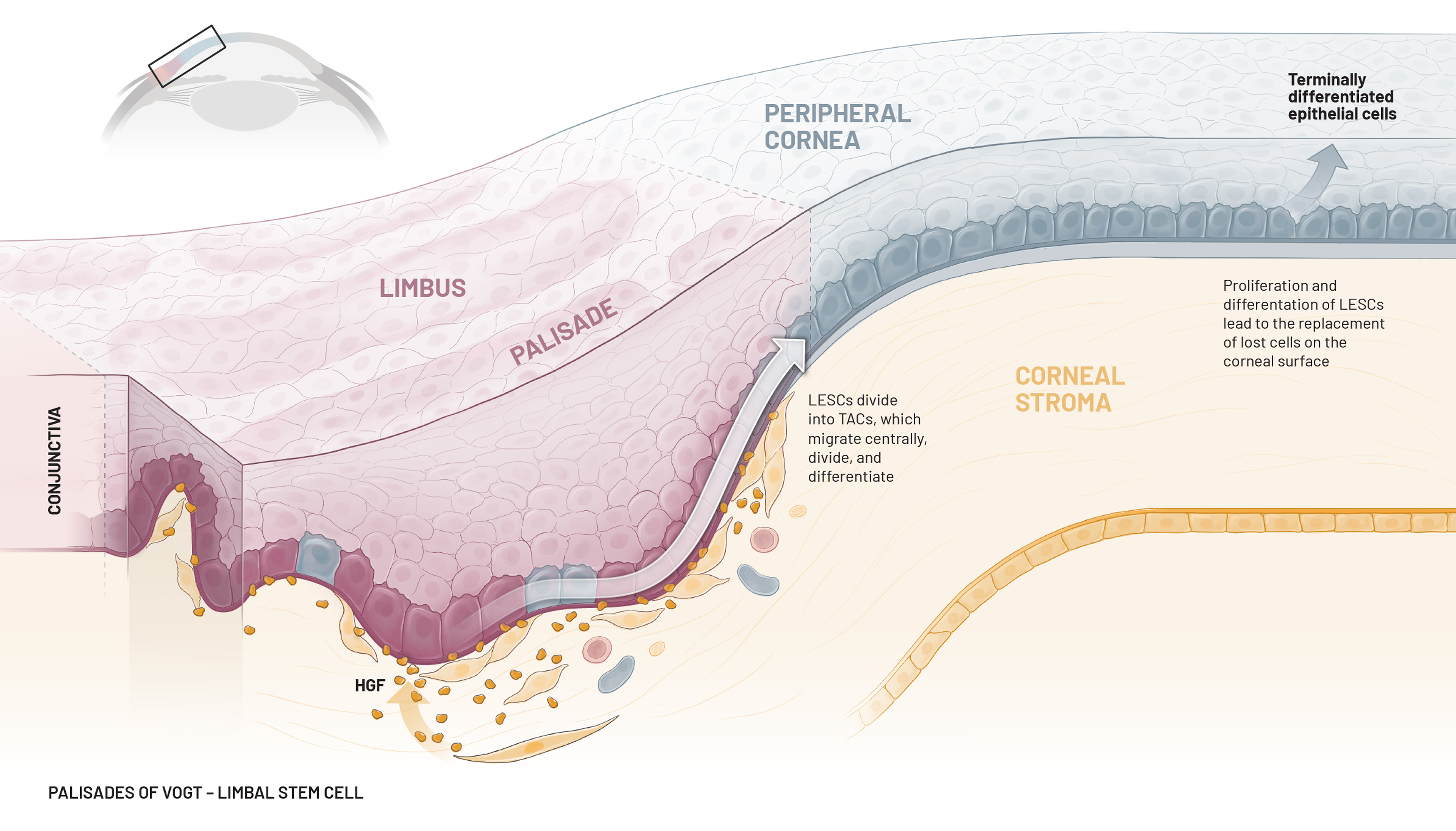

LESC

TAC

MSC

Swipe

The Unmet Need in LSCD
Limbal stem cell deficiency (LSCD) is a rare, progressive disease impacting the ocular surface, driven by underlying limbal stem cell dysfunction or direct injury. Loss or dysfunction of these stem cells, which are essential for regenerating and maintaining a stable, healthy corneal epithelial surface, can lead to severe vision loss and functional blindness.
Patients with LSCD experience chronic and debilitating symptoms, which most commonly include blurred vision, along with potential ocular discomfort or pain, photophobia, glare, and persistent epithelial defects that significantly impair daily activities and quality of life. LSCD is frequently under-recognized and misdiagnosed, with many patients initially treated for other corneal conditions, leading to delayed referral to specialists, inconsistent management, and disease progression before appropriate intervention.
Despite its profound impact on patients’ lives, there are currently no approved pharmaceutical therapies that address the underlying biology of LSCD. Today, disease management relies on long-term supportive care and, in advanced cases, complex surgical procedures that are resource-intensive, variably effective, and limited to specialized centers. As a result, many patients continue to experience ongoing disease progression and vision loss.
This significant unmet need underscores the importance of developing pharmacologic approaches designed to intervene earlier and address the fundamental drivers of LSCD.